Abstract
Objectives
To determine whether the lack of a Drug Enforcement Administration (DEA) registration is associated with differences in the treatment of pain for patients seen by Physician’s Assistants (PAs).
Methods
We conducted a retrospective cohort study of all ED patients seen by PAs over an 18 month period with a predefined list of diagnoses likely to need administration and/or prescription of analgesics. The primary outcome measurement was the proportion of patients administered or prescribed controlled substances. Other outcomes included prescriptions given by those PAs that obtained registration during the study, prescriptions given to patients that received controlled analgesics while in the ED, and the proportion of patients treated with muscle relaxants. Outcome comparisons were made between PAs with and without DEA registration.
Results
Of 4,386 charts reviewed, 40.7% of patients received a prescription for a controlled analgesic by those PAs with DEA registration vs. 28.4% of patients treated by PAs without DEA registration (Odds Ratio [OR] 1.72, 95% Confidence Interval [CI] 1.27 to 2.33). Respective proportions for benzodiazepines were 2.0% and 0.4% (OR 4.9, 95% CI 2.0-12.2).The proportions of patients receiving controlled substances while in the ED, where DEA registration is not required, were similar.
Conclusions
Patients treated for certain painful conditions in the ED were more likely to be discharged with a prescription for a controlled substance analgesic when seen by a PA with DEA registration as compared to a PA who did not possess a DEA registration.
Introduction
Common non-urgent medical conditions are frequently treated by Physician Assistants (PAs) in the Emergency Department (ED) setting. Over the past 10 years the number of PAs eligible to practice clinically has more than doubled and as of the beginning of 2008, there are 68,124 PAs in clinical practice. 1 It has become more common to have PAs working in EDs and practice arrangements vary. According to the 2008 census of the American Academy of Physician Assistants, 10% of all PAs who responded to the questionnaire practice clinically in an emergency department. 1 2
In our ED the PAs operated autonomously and consulted with a physician at their discretion. We had the opportunity to examine what medications patients were administered and prescribed for certain painful conditions that were unlikely to require physician consultation.
In the United States, Drug Enforcement Agency (DEA) registration is required for practitioners to write prescriptions for most controlled substances. DEA eligibility and requirements are influenced by federal, state and hospital rules and regulations. During the study period, PAs in New York State and several other states could obtain DEA registration and write prescriptions for controlled substances Schedules III, IV and V. State regulations do not generally require PAs to obtain DEA registration. The PA’s supervising physician generally determines if a DEA registration is required.
In our Emergency Department Physician Assistants were not required to have a DEA registration. If a PA does not have DEA registration then they must consult another practitioner with a valid DEA registration to generate a prescription for a controlled substance for a given patient. PAs in our hospital can administer controlled substances in the Emergency Department using the hospital’s DEA registration.
The literature presents multiple examples where patients with painful conditions are often undertreated for their pain. 3 4 5 6 7 Undertreatment may be due to minor impediments such as asking another provider to write a prescription.
We hypothesized that patients treated for certain painful conditions in the ED were more likely to be discharged with a prescription for a controlled substance when seen by a PA with a DEA registration as compared to a PA who did not possess a DEA registration.
Materials and Methods
Study Design
This was a retrospective cohort study based on medical chart review of patients seen in the hospital’s Fast Track. The independent or predictor variable was the DEA status of the treating PA and the outcome variables were the medication that the patient was administered in the ED and the prescription that the patient received. The hospital’s institutional review board approved the study protocol.
Study Setting and Population
A suburban academic ED in which the Fast Track treated approximately 27,000 patients of the total 88,000 annual census. The Fast Track is adjacent to the main ED. The PAs autonomously evaluated, treated and discharged patients without a physician seeing the patient unless they required contemporaneous physician involvement.
Study Protocol
We conducted a chart audit on patients seen by PAs in our Fast Track area for certain painful conditions (Table 1) that were selected a priority via consensus of the study authors, based on conditions that were likely to require a treatment for pain and were unlikely to have been seen by a physician. Seventy-five diagnoses were chosen that had 54 ICD-9 codes (specific diagnoses are available as a web appendix). The Fast Track was staffed by PAs with and without DEA registration. The PAs were not aware of the study. Data were collected retrospectively from 40,393 Fast Track visits (of 130,773 ED visits) during the 18-month study period.
All visits where PAs saw patients with any of the diagnoses chosen were studied. Data was abstracted from the electronic medical record (Allscripts ED™- formerly Healthmatics A4™, Chicago, IL). This computerized patient charting and order entry system enabled the collection of standardized information for each patient and integrated that information into a relational database. The SQL (structured query language) database was queried by Cognos Impromptu™ (IBM, Burlington, MA), which allows the administrator to create reports using filter criteria. Using the inclusion criteria listed below, a report was created with Cognos that queried all patients seen in the Emergency Department (4683) that fit the criteria for study enrollment. This report was further analyzed by Microsoft Excel 2007(Microsoft Excel, Redmond, WA) using tools that Cognos does not possess (Figure 1).
Every patient who was seen in the Fast Track area and discharged from July 1, 2005 to December 31, 2006 with a primary diagnosis listed in Table 1 was included in the study. There were no visits that were excluded, as there was 100% capture of data, which is characteristic of utilizing the electronic medical record as an inclusive database.
The data extracted about these patients included: arrival date/time; unique account number; age; sex; ethnic group; primary diagnosis; medications administered in the ED; text of each prescription; and the PA who saw each patient. The data on each PA was collected from administrative files and included their age, years of experience and employment, and the date they obtained DEA registration.
Outcome Measures
We assessed the association between the DEA status of the treating PA with the medications administered while in the ED and prescriptions written at discharge. We did not analyze for OTC recommendations.
Pain medications were classified as:
- Non-controlled pain medications (e.g., NSAIDs and tramadol)
- Controlled analgesics (opioids)
- Non-controlled muscle relaxants (e.g., cyclobenzaprine, methocarbamol)
- Controlled muscle relaxants (benzodiazepines)
Data Analysis
Microsoft Excel and SPSS 16.0 were used to analyze the DEA status of the treating PA and all subject characteristics and outcome measures. Comparisons of prescribing patterns were performed using logistic regressions utilizing generalized estimating equations in order to adjust for clustering effects of PAs and account for changes in PA status (PAs who obtained DEA registration during the study period). Odds Ratios (ORs) and 95% confidence intervals (CIs) were obtained from the generalized estimating equation results for the study hypothesis. All subgroup analyses used 99% CIs.
Results
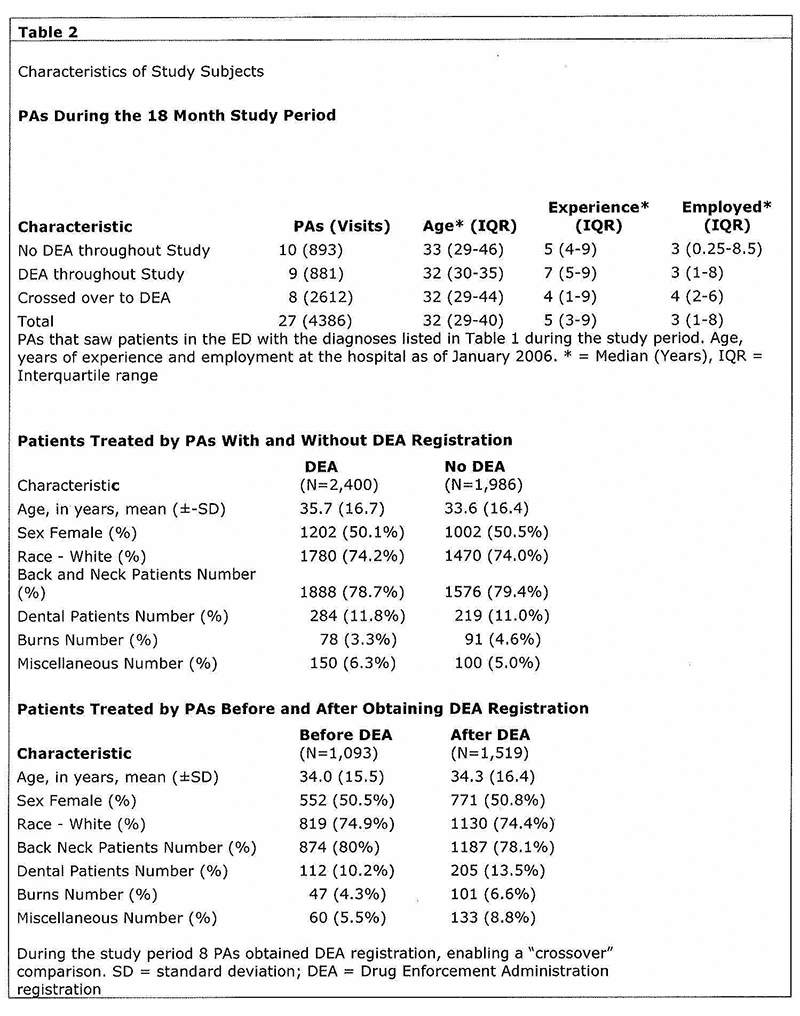
From 40,393 Fast Track visits, there were 4386 visits that were met study criteria. An overview of the 27 PAs and the 4,386 patient visits seen by PAs with and without DEA registration is shown in Figure 1. [fig. 1] The characteristics of the PAs and patients they treated are listed respectively in Table 2. [fig. 6] During the 18 month study period there were no significant differences in the groups and therefore they were considered comparable.
Primary Outcome
There were 2,400 patients seen by 18 PAs with a DEA registration at the time of the visit and 1,986 patients seen by 17 PAs that did not have a DEA registration at the time of the visit. Eight PAs obtained DEA registration during the study period.
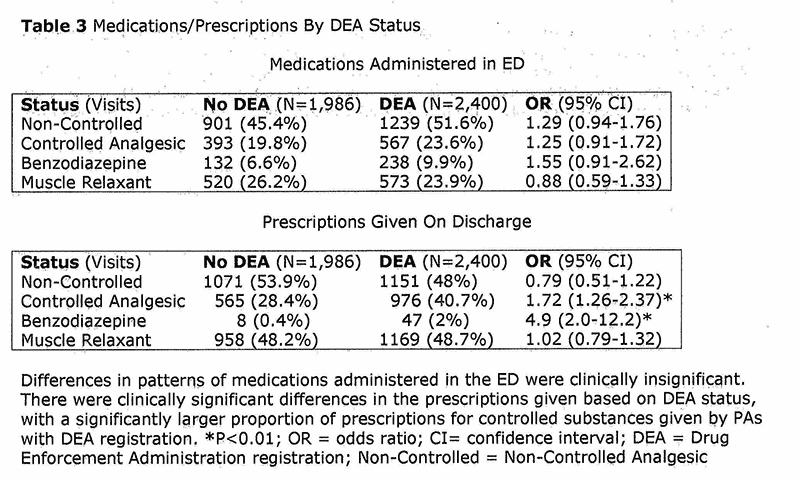
In the ED PAs with or without DEA registration could administer any controlled or non-controlled substance as they were administering medications under the hospital’s DEA registration. While patients were in the ED there were no clinically or statistically significant differences in medications administered by PAs with and without DEA registration. (Table 3) [fig. 8]
Patients were more likely to be discharged with a prescription for a controlled substance analgesic when seen by PAs with a DEA registration (40.7%) as compared to those PAs who do not possess a DEA registration (28.4%), (OR 1.72, 95% CI 1.26 to 2.37) Correspondingly, there was a statistically insignificant trend for patients to receive fewer prescription for a non-controlled analgesic (48% vs. 53.9%, OR 0.79, 95% CI 0.51-1.22). Patients were also more likely to be discharged with a prescription for a benzodiazepine when seen by PAs with a DEA registration as compared to those PAs who did not possess a DEA registration (2.0% vs.0.4%, OR 4.9, 95% CI 2.0-12.2).
Patients Treated by PAs Who Obtained Registration During the Study (Crossover Group)
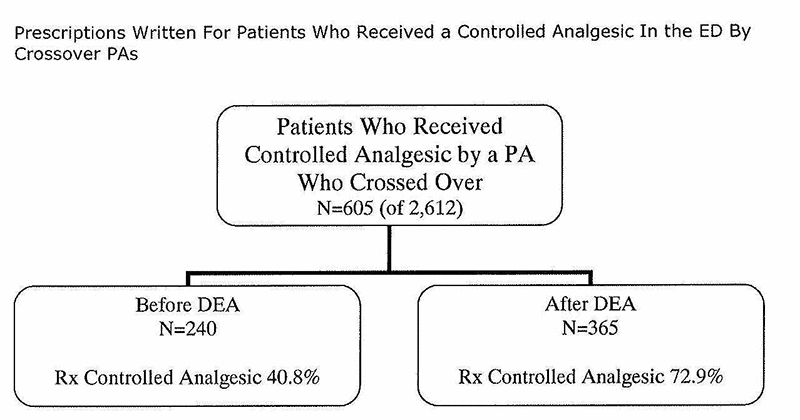
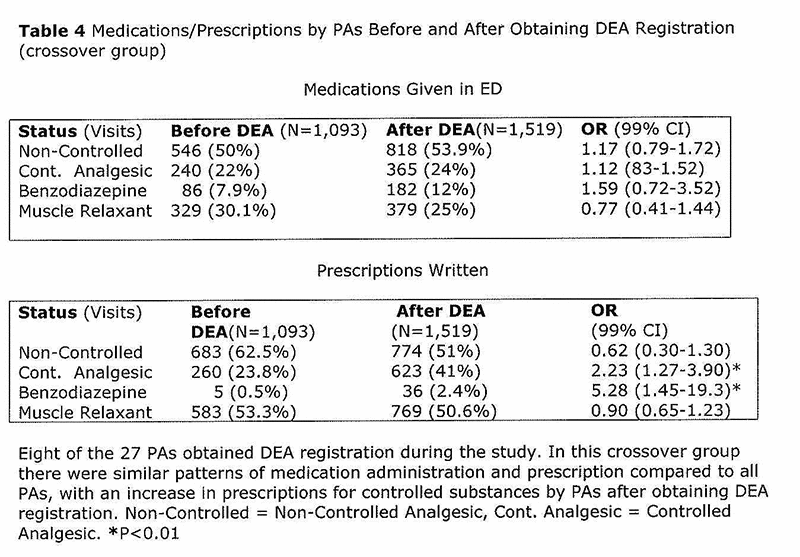
During the study period eight PAs obtained DEA registration and had 2,612 patient encounters, with a total of 1,093 encounters before and 1,519 after obtaining DEA registration. The proportions of pain medications/muscle relaxants given to these patients while in the ED by PAs before and after obtaining DEA registration during the study period are listed in Table 4. [fig. 9] There was no significant difference in medications administered while in the ED. However, there were increases in the proportions of patients receiving pain medication and muscle relaxant prescriptions from PAs after they obtained DEA registration during the study period. After PAs obtained DEA registration they were more likely to prescribe a controlled analgesics than before (41% vs. 23.8%, OR 2.23, 99% CI 1.27 to 3.90) and also more likely to generate a prescription for a benzodiazepine (2.4% vs. 0.5%, OR 5.28, 99% CI 1.45 to 19.29). Correspondingly, there was a trend to prescribe less non controlled analgesics after obtaining a DEA as compared to before (62.5% vs. 51%, OR 0.62, 99% CI 0.30-1.30). An overview of the 2,612 patients treated by the 8 PAs who crossed over is depicted in Figure 3. [fig. 3]
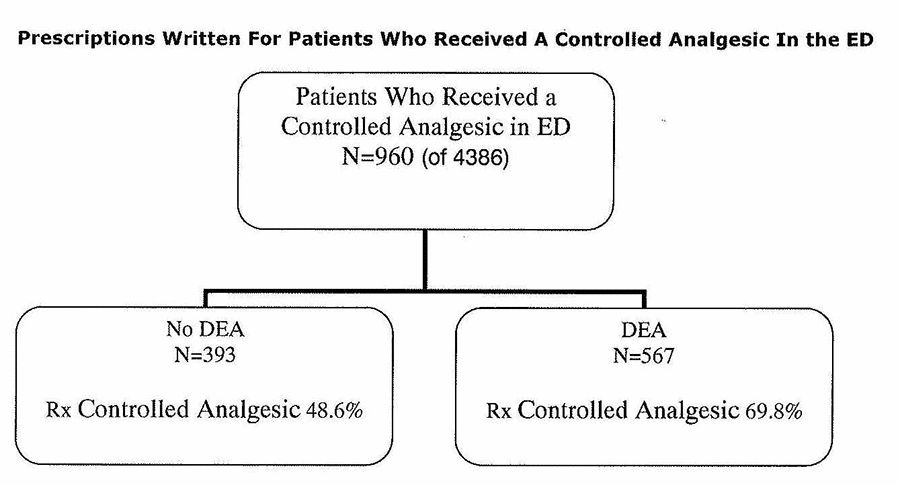
Patients Who Received Controlled Analgesics While in the ED
While in the ED 960 patients from the study population received a controlled analgesic (Figure 4). [fig. 4] Of these patients, 587 also received a prescription for a controlled analgesic. There was a larger absolute difference in prescriptions between those patients seen by PAs with DEA registration versus those without DEA registration 69.8% vs. 48.6% (OR 2.45, 99% CI 1.40 to 4.28). Of these 960 patients that received a controlled analgesic in the ED, 605 were seen by PAs who crossed over to obtain DEA registration. Figure 5 illustrates these patients and the proportion of controlled analgesic prescriptions written for these patients. The proportion of patients receiving a controlled analgesic prescription increased from 40.8% to 72.9% (OR of 3.89, 99% CI 2.15 to 7.05)
Discussion
As one of the most common presenting complaints, pain is often underestimated in the ED by healthcare providers. 8 9 It is important for providers of emergency care to adequately recognize and address a patient’s pain level satisfactorily. Even when a patient’s pain is appropriately recognized it is often not properly treated. 10
There has been a trend in Emergency Departments to create Fast Tracks staffed by PAs and other mid-level providers. 11 12 In many instances, these providers are not required to have DEA registration, but are permitted to administer controlled substances to patients for pain while in the ED under the aegis of the hospital’s DEA registration. However, in order for a PA to generate a prescription for a controlled substance, they must either have their own DEA registration, or obtain the prescription from a healthcare provider who does have a DEA registration. We found no difference in the type of medication administered to patients while they were in the ED where no DEA registration is required.
The change in our primary outcome (prescriptions for controlled substances generated) can be explained by differences in DEA status of our independent variable (the DEA status of the treating PA). The results support our hypothesis that there is an association between the DEA registration status of PAs and the prescriptions their patients receive for controlled substances. We found that PAs with DEA registration generated more prescriptions for controlled substances than those without DEA registration. Correspondingly, there was also a statistically insignificant trend for those PAs with DEA registration to generate fewer prescriptions for non-controlled substances.
During the study period some of the PAs obtained DEA registration. This enabled us to analyze the association between change in DEA registration status and prescribing patterns. This crossover sub-group analysis reinforces our hypothesis of the association between current DEA status and prescriptions written for certain painful conditions.
A more clinically relevant outcome would be to look at what prescriptions patients received at discharge when they were administered a controlled analgesic while in the ED. It can be assumed that if a patient received a controlled analgesic while in the ED, then they would more likely benefit from a prescription for a controlled analgesic at discharge. We looked at patients who received a controlled analgesic while in the ED, and their prescriptions at discharge. These patients were more likely to be prescribed a controlled analgesic at discharge. In this subgroup there was an even stronger association between having a DEA registration and the likelihood of a patient receiving a prescription for a controlled analgesic.
There was a trend for PAs without DEA registration to prescribe more non-controlled analgesics. It could be argued that the inconvenience of having to ask another provider to write a prescription for a controlled medication sometimes resulted in the substitution of a non-controlled analgesic for a controlled analgesic (that they would otherwise have received) and thus some patients were deprived of an appropriate analgesic prescription for the selected painful conditions. This trend for substituting non-controlled for controlled prescriptions could account for much of the difference in non-controlled substances seen between the PA groups. It would require a larger group of PAs and patients to conclude that there was a significant pattern to substituting non-controlled prescriptions for controlled substances by PAs without a DEA registration.
Conclusions
Patients treated for certain painful conditions in the ED are more likely to be discharged with a prescription for a controlled substance when seen by PAs with a DEA registration as compared to those PAs who do not possess a DEA registration. There is a strong association between the prescriptions patients receive for certain painful conditions and whether the treating PA has DEA registration. PAs who obtained DEA registration markedly changed their prescribing habits by writing more prescriptions for controlled substances in the treatment of these painful conditions. Further studies should be done to assess whether patients who receive prescriptions for controlled substances have improved management of their pain and whether midlevel practitioners who treat similar painful conditions should be required to have DEA registration.
Acknowledgments: we wish to thank Curt Cicotte for his data collection
Figures & Tables
-
Flow of visits that were analyzed during the study period. There were 4386 visits analyzed according to the DEA status of the PA that treated each patient. SQL = structured query language, PA = Physician Assistant, DEA = Drug Enforcement Administration
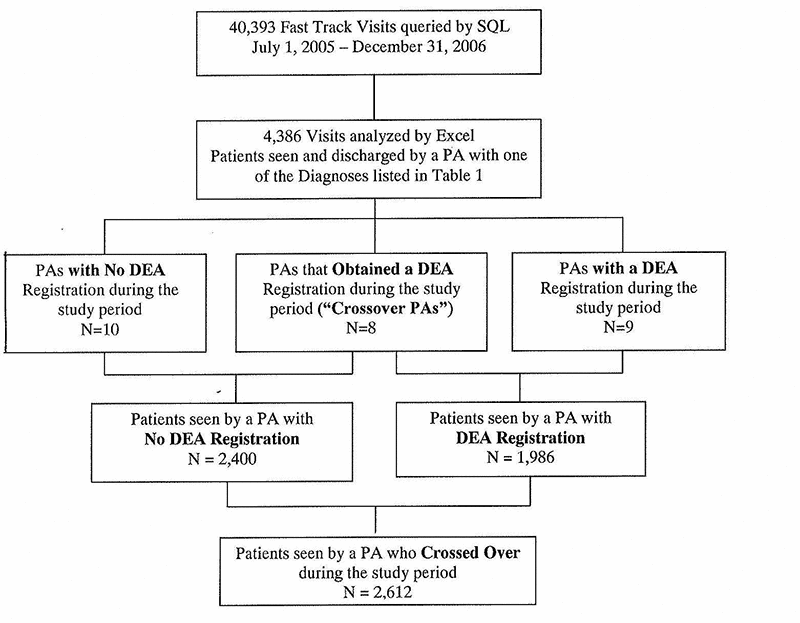
-
Of the 4,386 charts audited, 1986 patients were seen by a PA without DEA registration (N-18) and 2,400 patients were seen by a PA with DEA registration (N=17). On discharge from the ED, 28.4% of patients seen by a PA without DEA registration were given a prescription for a controlled analgesic, while 40.7% of patients who were seen by a PA with DEA registration were given a prescription for a controlled analgesic (OR 1.72, 95% CI 1.26 to 2.37) and correspondingly more benzodiazephines were prescribed (2.0% vs. 0.4%%, OR 4.9, 99% CI 2 to12.2).

-
During the 18 months of observation, 8 of 27 PAs obtained DEA registration, allowing a before and after comparison. More controlled analgesics were prescribed once DEA registration was obtained (41% vs. 23.8%, OR 2.23, 99% 1.27 to 3.90) and correspondingly more benzodiazephines were prescribed (2.4% vs. 0.5%, OR 5.28, 99% CI 1.45 to 19.3). DEA – Drug Enforcement Administration registration, PA = Physician Assistant, Rx = prescriptions generated

-
A subgroup of 960 patients receive a controlled analgesic while in the ED. PAs with DEA registration were more likely to prescribe a controlled analgesic to these patients compared to those PAs without DEA registration (OR 2.45, 99% CI 1.40 to 4.28). DEA = Drug Enforcement Administration registration, PA = Physician Assistant, Rx = prescriptions generated
-
A subgroup of 605 of 960 patients was administered controlled analgesics in the ED by the 8 PAs who obtained DEA registration during the course of the study. In this crossover subgroup, more prescriptions for controlled analgesics were written for these patients by PAs after they obtained DEA registration as compared to the period before the PAs obtained DEA registration (OR 3.89, 99% CI 2.15 to 7.05).
-

-
-
-
-

References
-
American Academy of Physician Assistants.2008 AAPA Physician Assistant Census Report. http://www.aapa.org/research/Highlights08/2008AAPACensusNationalReport.pdf Accessed March 3, 2009.
-
American Academy of Physician Assistants.1998 Physician Assistant Census Report. http://www.aapa.org/research/sum98.html. Accessed March 3, 2009.
-
Wilson JE, Pendleton JM. Oligoanalgesia in the emergency department. Am J Emerg Med. 1989;7:620-623
-
Stalnikowicz R, Mahamid R, Kaspi S, Brezis M. Undertreatment of acute pain in the emergency department: a challenge. Int J Qual Health Care. 2005;17:173-176.
-
Brown JC, Klein EJ, Lewis CW, Johnston BD, Cummings P. Emergency department analgesia for fracture pain. Ann Emerg Med. 2003;42:197-205.
-
Rupp T, Delaney KA. Inadequate analgesia in emergency medicine. Ann Emerg Med. 2004;43:494-503.
-
Grant PS. Analgesia delivery in the ED. Am J Emerg Med. 2006;24:806-809.
-
Neighbor M, Puntillo, KA, O’Neil, N. Nurse underestimation of patient pain intensity. Society for academic emergency medicine. 1999;6:514
-
Guru V, Dubinsky I. The patient vs. caregiver perception of acute pain in the emergency department. J Emerg Med. 2000;18:7-12.
-
Ducharme J, Barber C. A prospective blinded study on emergency pain assessment and therapy. J Emerg Med. 1995;13:571-575
-
Counselman FL, Graffeo CA, Hill JT. Patient satisfaction with physician assistants (PAs) in an ED fast track. Am J Emerg Med. 2000;18:661-665.
-
Counselman FL, Schafermeyer RW, Perina DG. Academic departments of emergency medicine: the effects of managed care. Acad Emerg Med. 1998;5:1095-1100.
-
Sigler KA, Guernsey BG, Ingrim NB, et al. Effect of a triplicate prescription law on prescribing of Schedule II drugs. Am J Hosp Pharm. 1984;41:108-111.
-
Simoni-Wastila L, Ross-Degnan D, Mah C, Gao X, Brown J, Cosler LE et al. A retrospective data analysis of the impact of the New York triplicate prescription program on benzodiazepine use in medicaid patients with chronic psychiatric and neurologic disorders. Clin Ther. 2004;26:322-336.
-
McGerald G, Dvorkin R, Levy D, Lovell-Rose S, Sharma A. Prescriptions for schedule II opioids and benzodiazepines increase after the introduction of computer-generated prescriptions. Acad Emerg Med. 2009;16:508-512.
